Research
Basic Properties
Empirical and Molecular Formula- H2CO3
Molar Mass- 62.03 g/mole
Density- 1.668 g/cm3
Molar Mass- 62.03 g/mole
Density- 1.668 g/cm3
Research Narrative
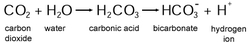
Chemical Reaction- Small traces of carbonic acid are formed when carbon dioxide is dissolved in water. H2CO3 usually exists as a solution. The chemical reaction is again seen to the right.
Molecular Structure- Carbonic acid possesses a non-crystalline structure. Three oxygen atoms are bonded to a central carbon atom, with two of the three oxygen atoms bonded to one hydrogen atom each. This molecule does not have a uniform distribution of negative charge, and it is, therefore, polar.
More Chemistry Explained Through Real World Application-
The final major property of carbonic acid is that it is (you guessed it!) an acid. Acids are defined as any substance that releases hydrogen ions into solutions, i.e. has a pH of less that 7. When excess hydrogen ions build up in the body, or the fluids become too acidic, then bicarbonate ions accept extra hydrogen ions and keeps the body’s pH at a normal level. However, if the hydrogen ion levels drop too much, or the fluids become too alkaline, carbonic acid gives up hydrogen ions. pH levels must be maintained to keep the body in a state of homeostasis, where processes can be carried of effectively and efficiently.
Synthesis and Isolation
Gas-phase carbonic acid instantly decomposes into water and carbon dioxide. It is only detectable as a trace component. However, carbonic acid has recently been isolated. Until recently, the molecule resisted all attempts to be isolated or detected.
Researchers demonstrated that carbonic acid may exist in the gas phase and that it is stable at temperatures up to –30 °C. In these experiments, solid carbonic acid was formed via acid-base reactions at very low temperatures and then warmed to –30 °C. Any evaporating molecules were contained in the noble gas argon and then quickly cooled again. This resulted in a "image" of gas-phase carbonic acid. Through infrared spectrometry, researchers could study the isolated molecule.
The resulting data was of great interest to astronomers, as this made it easier to detect gas-phase carbonic acid in space, where it is theorized to be present in the tails of comets and on the planet Mars. From this data, other more complex properties were identified, and the quantum structure of the molecule was better understood.
Disposal- As this chemical is not hazardous in any form (in fact, it is inside our bodies at this very moment!), there are no necessary or unique means by which to properly dispose of carbonic acid, or solutions of it.
Molecular Structure- Carbonic acid possesses a non-crystalline structure. Three oxygen atoms are bonded to a central carbon atom, with two of the three oxygen atoms bonded to one hydrogen atom each. This molecule does not have a uniform distribution of negative charge, and it is, therefore, polar.
More Chemistry Explained Through Real World Application-
The final major property of carbonic acid is that it is (you guessed it!) an acid. Acids are defined as any substance that releases hydrogen ions into solutions, i.e. has a pH of less that 7. When excess hydrogen ions build up in the body, or the fluids become too acidic, then bicarbonate ions accept extra hydrogen ions and keeps the body’s pH at a normal level. However, if the hydrogen ion levels drop too much, or the fluids become too alkaline, carbonic acid gives up hydrogen ions. pH levels must be maintained to keep the body in a state of homeostasis, where processes can be carried of effectively and efficiently.
Synthesis and Isolation
Gas-phase carbonic acid instantly decomposes into water and carbon dioxide. It is only detectable as a trace component. However, carbonic acid has recently been isolated. Until recently, the molecule resisted all attempts to be isolated or detected.
Researchers demonstrated that carbonic acid may exist in the gas phase and that it is stable at temperatures up to –30 °C. In these experiments, solid carbonic acid was formed via acid-base reactions at very low temperatures and then warmed to –30 °C. Any evaporating molecules were contained in the noble gas argon and then quickly cooled again. This resulted in a "image" of gas-phase carbonic acid. Through infrared spectrometry, researchers could study the isolated molecule.
The resulting data was of great interest to astronomers, as this made it easier to detect gas-phase carbonic acid in space, where it is theorized to be present in the tails of comets and on the planet Mars. From this data, other more complex properties were identified, and the quantum structure of the molecule was better understood.
Disposal- As this chemical is not hazardous in any form (in fact, it is inside our bodies at this very moment!), there are no necessary or unique means by which to properly dispose of carbonic acid, or solutions of it.
