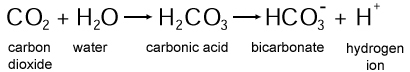Introduction
Fundamental Properties
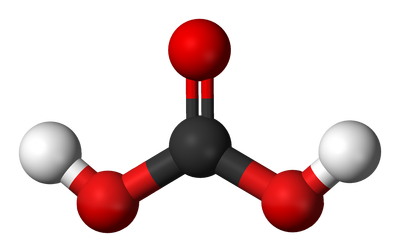
Carbonic acid is a weak acid that is created when carbon dioxide (CO2) reacts with water (H2O), resulting in the chemical formula H2CO3. A pure form of carbonic acid cannot be isolated as a solid or liquid, but rather as a gas. As its name suggests, carbonic acid is slightly acidic, meaning it has a pH value below 7.
Real World Applications
|
Geology- Limestone dissolves under the action of rainwater and groundwater, charged with carbonic acid, as well as naturally occurring organic acids. This leads to the formation of caves, as well as its stalactites and stalagmites.
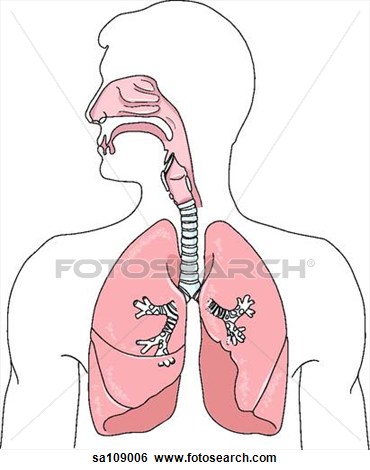
Food/Drink- Carbonic acid is present in frequently consumed carbonated drinks. Anatomy/Physiology- Carbonic acid is an important step in the transport of carbon dioxide out of the body via respiratory gas exchange. The CO2, a by-product of cellular respiration, reacts with water in order to move through the bloodstream and eventually to the lungs. It is described as respiratory acid. In addition, while carbonic acid travels through the bloodstream, it plays an essential role in pH regulation. Medicine- Carbonic acid is used topically for dermatitides, mouthwash, and veterinary use as an emergency emetic. |
Alexander Philips
Honors Chemistry, Period E
Mrs. Fontaine
5-1-15
Honors Chemistry, Period E
Mrs. Fontaine
5-1-15